A completely incisionless device that replicates metabolic surgery without making any cuts could surpass current technologies for managing metabolic conditions, providing unmatched treatment for millions of people who reject invasive surgery or do not respond to drugs.
In a study authored by an international team of prominent academics including scientists from King’s, researchers found that the ForePass endoscopic showed extraordinary efficacy in treating conditions such as severe obesity, type 2 diabetes, and metabolic dysfunction-associated steatohepatitis (MASH).
Published in the journal Gut, the study aimed to evaluate the ForePass device’s impact on weight, insulin sensitivity, and fecal microbiota (a range of microorganisms) in pigs, compared to a control group that did not undergo the procedure.
Results revealed a 79% reduction in weight gain compared to the control group, suggesting it may be substantially more effective than common metabolic surgeries like gastric bypass. It also showed substantial improvements in how the body handles insulin and reduces glucose in the liver compared to controls.
Finally, the study found markedly improved response to insulin, enhanced use of glucose, and beneficial changes in metabolic health-related gut bacteria.
“The ForePass device will be a game-changer for treating severe metabolic conditions. It replicates the effects of metabolic surgery without invasive procedures and without making any cuts to internal organs. It’s the first device of its kind and will pave the way for a new era in managing severe metabolic conditions,” said lead author Dr. Manoel Galvao Neto, endoscopic surgeon and researcher based at Sri Aurobindo Medical College, India.
Dr. Neto continued, “It is a crucial development, as only 1% of patients opt for bariatric surgery due to its invasive nature. The medical community is eagerly anticipating the upcoming clinical trials involving the ForePass device.”
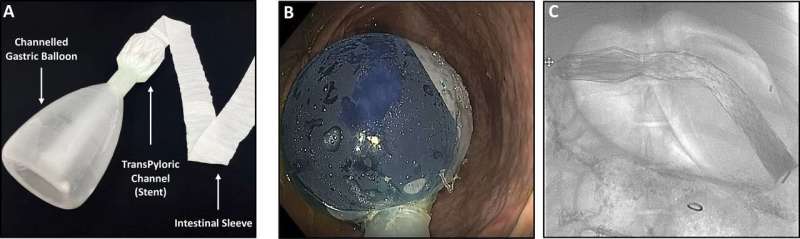
ForePass is an innovative device, developed by Keyron, that combines a gastric balloon crossed by a central channel that connects to a flexible intestinal sleeve, thereby effectively replicating the mechanism of invasive metabolic surgery without the need for surgery or making any incisions.
The device is inserted into the stomach and initial tract of the small intestine using endoscopy, a much less invasive and cheaper procedure compared to metabolic surgery. Unlike metabolic surgery, ForePass is fully reversible, making it an appealing option for patients.
For the study, the pigs were implanted with ForePass for a month, and the device demonstrated a significant 79% decrease in weight gain and marked enhancements in glucose balance compared to control animals who were not implanted with the device.
Additionally, it favorably altered fecal microbiota, boosting bacteria linked to metabolic well-being. These outcomes align with a reversal of severe obesity, diabetes, and MASH, indicating an overall improvement in metabolic health.
“This study’s findings are extraordinary and unprecedented. We learned that ForePass could be even more effective than metabolic surgery. Traditional treatments, including drugs, fall short for individuals with higher BMIs. ForePass stands out as it offers the benefits of metabolic surgery without the need for actual surgery, making it a game-changer for millions who are reluctant to undergo surgery. It promises to revolutionize treatment for severe obesity and insulin resistance-related pathologies like diabetes and various liver diseases,” said Co-author Geltrude Mingrone, Professor of Diabetes and Nutrition, King’s.
ForePass is designed to decrease food consumption, resulting in considerable weight loss, and directs food deeper into the gut. This process significantly improves insulin resistance, effectively treating or potentially reversing diabetes and serious liver conditions such as MASH.
“Considering that existing medications fall short in achieving the necessary weight loss for individuals with very high BMIs, ForePass will be a transformative solution for these patients. It has the potential to significantly impact the lives of tens of millions of people in the U.S. and EU alone,” said Co-author Professor Stefan R Bornstein, Chair in Diabetes and endocrinology consultant based at King’s.
Keyron plans to initiate clinical trials for ForePass, which is anticipated to start in early 2025. Should clinical trials confirm the safety and efficacy seen in animals, the device has the potential to revolutionize the treatment of severe obesity over the coming decades.
More information:
Giulia Angelini et al, ForePass endoscopic bypass device for obesity and insulin resistance—metabolic treatment in a swine model, Gut (2023). DOI: 10.1136/gutjnl-2023-331335
King’s College London
Citation:
Incisionless device could revolutionize treatment for diabetes, liver disease and severe obesity (2024, January 19)
retrieved 19 January 2024
from https://medicalxpress.com/news/2024-01-incisionless-device-revolutionize-treatment-diabetes.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
_______
Source : medicalxpress.com